 PDF(4040 KB)
PDF(4040 KB)


 PDF(4040 KB)
PDF(4040 KB)
 PDF(4040 KB)
PDF(4040 KB)
不同pH的碱性环境中16Mn钢及热影响区应力腐蚀行为
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Stress Corrosion Cracking Behavior of 16Mn Steel and Heat-affected Zone in Alkaline Sulfide with Different pH Value
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}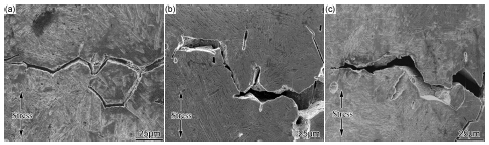
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |