 PDF(7267 KB)
PDF(7267 KB)


 PDF(7267 KB)
PDF(7267 KB)
 PDF(7267 KB)
PDF(7267 KB)
添加剂辅助水热热解制备尺寸可控纳米孔碳微球
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Additive-assisted hydrothermal route to synthesize nanoporous carbon microspheres with controlled particle size
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}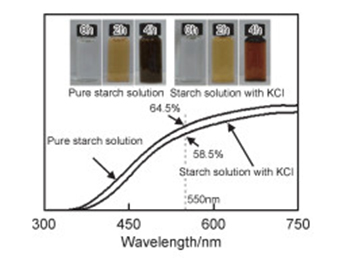
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |