 PDF(11838 KB)
PDF(11838 KB)


 PDF(11838 KB)
PDF(11838 KB)
 PDF(11838 KB)
PDF(11838 KB)
超薄材料电催化CO2还原合成液体燃料
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Ultra-thin materials for electrocatalytic CO2 reduction to prepare liquid fuels
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}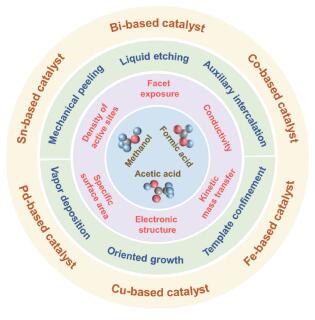
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |