 PDF(12462 KB)
PDF(12462 KB)


 PDF(12462 KB)
PDF(12462 KB)
 PDF(12462 KB)
PDF(12462 KB)
Ⅰ型胶原/海藻酸钠/透明质酸复合水凝胶用于血管组织工程细胞负载与3D培养
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Type Ⅰ collagen/sodium alginate/hyaluronic acid composite hydrogel for cell loading and 3D culture in vascular tissue engineering
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}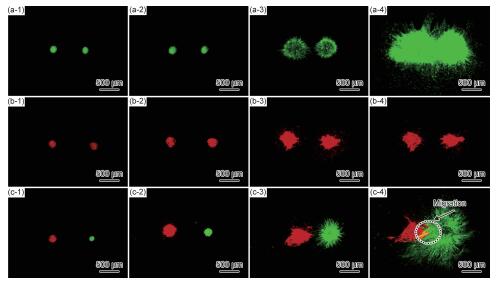
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |