 PDF(3279 KB)
PDF(3279 KB)


合金元素配分对珠光体相变热动力学及其奥氏体化影响的研究进展
杨泽南, 李赛, 于俊杰, 谢强, 王祯, 张明达, 董浩凯, 张强, 杨志刚
材料工程 ›› 2020, Vol. 48 ›› Issue (7) : 61-71.
 PDF(3279 KB)
PDF(3279 KB)
 PDF(3279 KB)
PDF(3279 KB)
合金元素配分对珠光体相变热动力学及其奥氏体化影响的研究进展
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Progress in effect of alloying element partition on thermodynamics and kinetics of pearlite transformation and its austenitization
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}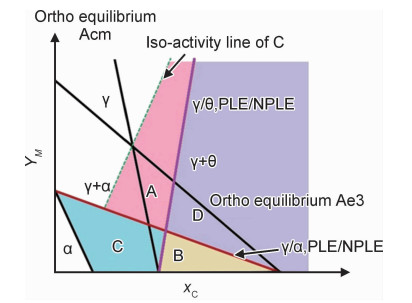
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |