 PDF(4574 KB)
PDF(4574 KB)


 PDF(4574 KB)
PDF(4574 KB)
 PDF(4574 KB)
PDF(4574 KB)
石墨烯的制备及其对环氧树脂导电性能的影响
 ({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}
({{custom_author.role_cn}}), {{javascript:window.custom_author_cn_index++;}}Preparation of Graphene and Its Effect on Conductivity Epoxy Resin
 ({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}
({{custom_author.role_en}}), {{javascript:window.custom_author_en_index++;}}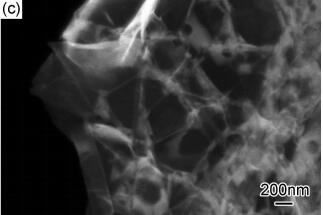
| {{custom_ref.label}} |
{{custom_citation.content}}
{{custom_citation.annotation}}
|
/
| 〈 |
|
〉 |